2025 has been quite a strange year for medicine in Canada. It’s one of those years when you might pop into a pharmacy for something simple. But then you find out about recalls that you perhaps weren’t expecting.
Here are the products that have been recalled & exactly why.
Cough syrup recall

Health Canada issued a Type II drug recall on December 5, 2025, for Life Brand’s Mucus & Phlegm Relief Extra Strength. The problem was that certain batches had been stored outside of their recommended temperatures, affecting their strength.
The affected batches were the 200 mg/5 ml varieties of 12487, 12525, 12684, 12530 & 12691. These were sold in Calgary.
Glucose sensors

Just two days before, on December 3, Canadian officials recalled Abbott Diabetes Care’s FreeStyle Libre 3 Plus Sensor Kit. It was a Type II recall.
Some of the sensors on the T60003522 & T60003216 lots apparently produced the incorrect low readings. Officials recommended users stop using any affected products.
Nitrous oxide chargers

The Canadian government issued a Type I recall on November 10 for numerous nitrous oxide chargers under the brands Bamboozle, Best Whip, Gold Whip, GreatWhip, Prime Whip & Whip-It!.
These were sold for inhalation, although none of these included a DIN. They were recalled, ironically, for being sold without proper authorization for inhalation.
Hospital protective gear
Hospital facilities have also suffered recalls. The government recalled ChemoPlus sleeves & gowns from Cardinal Health on December 3rd after it emerged that they were printed with five-year expiry dates instead of the correct three years.
The recall covered numerous sizes and was for both sleeves & open-back or close-back gowns.
Surgical tools

Zimmer’s Air Dermatome & Dermatome AN handpieces also received a Type II recall on the same day. The recall came from five complaints due to the thickness control bar being misaligned, which caused cutting depth problems.
It covered the handpieces with the serial numbers 88710100 and 00-8801-001-00.
Wound dressings
Late August 2025 saw Medihoney Antibacterial Dressings being recalled. It emerged that there were possible breaches in the sterile seal.
All lots with the catalogue numbers 391, 395, 799, 784, 781, 780, 785, 398, 782, 787 & 783 were affected. The recall came after earlier health warnings about weak seals and issues with the twist-off cap.
Unauthorized libido enhancement products

Numerous unauthorized enhancement products went onto Health Canada’s advisory list in July 2025. You could find many of these items in gas stations & corner stores.
Testing revealed that these products included undeclared prescription ingredients like sildenafil. Some of the affected products were Bang Maximum Power and Rhino 7 Platinum 500.
Contact lenses
Alcon’s PRECISION1 one-day lenses were given a Type II recall on October 7th. It came after testing found that some of the units within the A8041128 lot could have a weak seal.
Officials confirmed that the units were currently sealed, but they issued the recall after concerns over how well the seal would perform over the lenses’ shelf life.
Blood pressure medicine

Government officials issued a recall of Amlodipine 10 mg tablets in September because it emerged that a few batches had tablets that were larger than they should’ve been.
Such an issue was classified as a product quality issue. The affected tablets were DIN 02429225 with a lot number of 2403038A.
Birth control packs
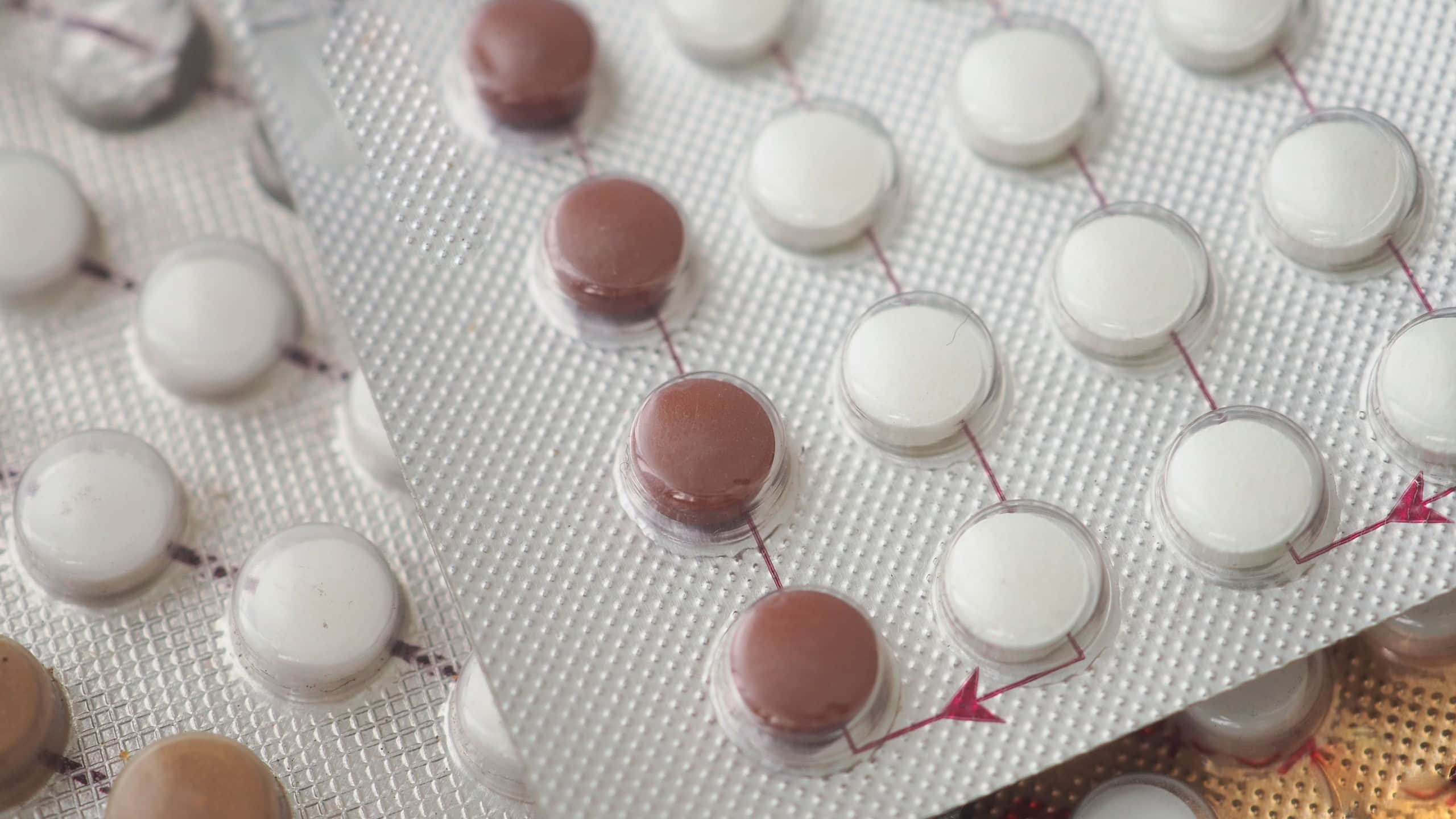
Teva Canada reported packaging mix-ups earlier in the year, leading to a recall of Seasonale & Seasonique in July 2025.
Two of the Seasonale lots (100059661 & 100069150) had an extra row of placebo pills. The Seasonique lot (100069151) was recalled because of a possible extra row of tablets in the first two trays.
Sources: Please see here for a complete listing of all sources that were consulted in the preparation of this article.